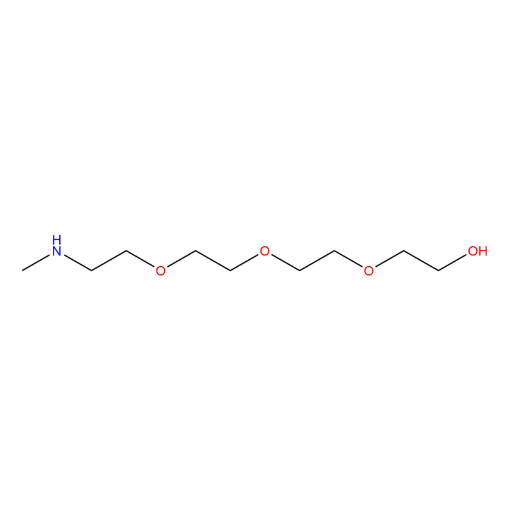
Name: 5,8,11-Trioxa-2-azatridecan-13-ol
Molecular Formula: C9H21NO4
CAS#: 90430-59-4
SMILES: OCCOCCOCCOCCNC
MDL#: MFCD29042347
Catalog#: AMTGC828-HA21
Molecular weight: 207.27 g/mol
Other names:
- HYDROXY-PEG3-METHYLAMINE
- Amino-PEG3-alcohol
- PEG3 amino alcohol
- Hydroxy-PEG3-amine
Fields of Interest: bioconjugation, materials science, and drug delivery
Background & Applications:
Background
5,8,11-Trioxa-2-azatridecan-13-ol (CAS 90430-59-4) is a heterobifunctional amine-functionalized PEG derivative based on a PEG3 backbone, featuring a terminal primary alcohol and an internal secondary amine. The polyethylene glycol spacer provides hydrophilicity, flexibility, and compatibility with aqueous and organic environments, while enabling defined linker length for spatial control. The hydroxyl group offers opportunities for derivatization through esterification, ether formation, or activation to leaving groups, while the secondary amine provides tunable reactivity and basicity for selective transformations. This compound serves as a versatile intermediate within a comprehensive portfolio of functionalized PEGs designed for modular and controlled molecular modification.
Applications
5,8,11-Trioxa-2-azatridecan-13-ol is commonly used in bioconjugation, materials science, and drug delivery applications where dual functionality and PEG spacing are required. Typical uses include stepwise synthesis of multifunctional linkers, PEGylation of small molecules or polymers, and preparation of intermediates for pharmaceutical and biomaterials research. The combination of a reactive hydroxyl group and a secondary amine enables orthogonal or sequential functionalization strategies. As part of a robust functionalized PEG product line, this PEG3 amino-alcohol supports modular design strategies in specialty chemical synthesis, diagnostics, and advanced materials development.
Appearance: Colorless liquid
Purity: 98%
Storage: -20 °C
Solubility: Dichloromethane, Chloroform, Methanol
Literature:
- Journal of Medicinal Chemistry, 2023, vol. 66, # 14, p. 9445 – 946