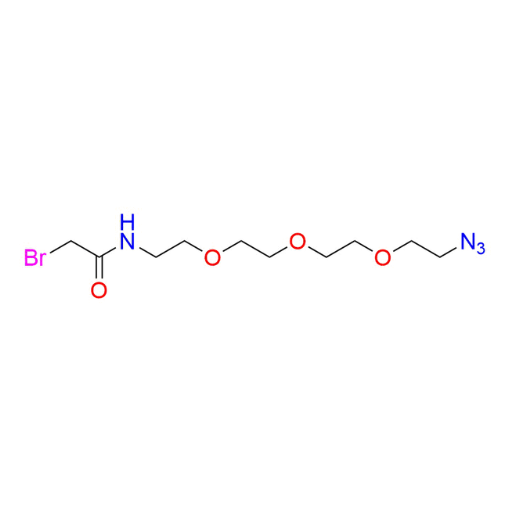
Name: N-(2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)ethyl)-2-bromoacetamide
Molecular Formula: C10H19BrN4O4
CAS#: 940005-81-2
SMILES: O=C(CBr)NCCOCCOCCOCCN=[N+]=[N-]
MDL#: MFCD22683302
Catalog#: AMTH395-BA24
Molecular weight: 373.42 g/mol
Other names:
- Azido-PEG4-bromoacetamide
- PEG4 azide bromoacetamide linker
- Thiol-reactive azido PEG linker
Fields of Interest: bioconjugation, chemical biology, and materials science
Background & Applications:
Background
N-(2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)ethyl)-2-bromoacetamide is a heterobifunctional azide-functionalized PEG conjugate based on a PEG4 backbone, featuring a terminal azide group and a bromoacetamide electrophile. The polyethylene glycol spacer provides hydrophilicity, flexibility, and compatibility with aqueous and biological environments while enabling defined linker length for spatial separation. The azide functionality supports efficient bioorthogonal click chemistry, including CuAAC and strain-promoted azide–alkyne cycloaddition, while the bromoacetamide group is a well-known electrophile for selective reaction with thiols, particularly cysteine residues in biomolecules. This dual-reactive architecture makes the compound a versatile intermediate within a comprehensive portfolio of functionalized PEGs designed for orthogonal and site-specific molecular assembly.
Applications
This azido-PEG4–bromoacetamide linker is commonly used in bioconjugation, chemical biology, and materials science applications where selective thiol reactivity and click chemistry are combined. Typical uses include cysteine-specific labeling of peptides and proteins via the bromoacetamide moiety, followed by click-based attachment to alkynyl-functionalized probes, polymers, or surfaces. It is also used in the preparation of multifunctional intermediates and PEGylated biomolecules. The combination of thiol-reactive electrophile and click-compatible azide enables highly controlled, stepwise conjugation strategies. As part of a robust functionalized PEG product line, this compound supports modular design strategies in diagnostics, biomaterials, and advanced chemical synthesis.
Appearance: Yellow Liquid
Purity: 98%
Storage: Room Temp
Solubility: Dichloromethane, Chloroform, Methanol
Literature:
- Journal of Medicinal Chemistry, 2021, vol. 64, # 16, p. 12359 – 12378
- Angewandte Chemie – International Edition, 2021, vol. 60, # 49, p. 25914 – 25921
- Bioorganic and Medicinal Chemistry Letters, 2007, vol. 17, # 9, p. 2459 – 2464