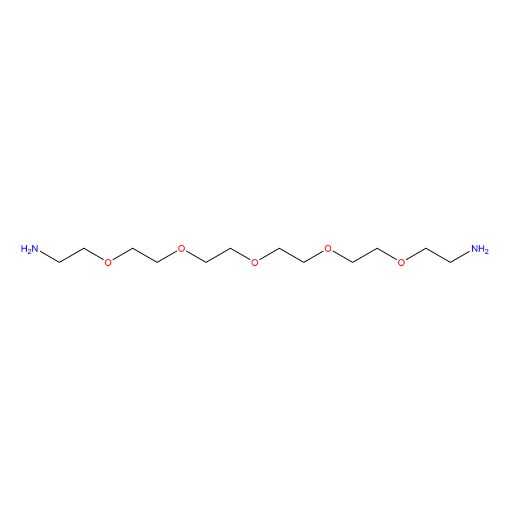
Name: 3,6,9,12,15-Pentaoxaheptadecan-1,17-diamine
Molecular Formula: C12H28N2O5
CAS#: 72236-26-1
SMILES: NCCOCCOCCOCCOCCOCCN
MDL#: MFCD07369626
Catalog#: AMTGC997-PD22
Molecular weight: 280.36 g/mol
Other names:
- 1,17-diamino-3,6,9,12,15-pentaoxaheptadecane
- PEG5 diamine
- α,ω-Diamino-pentaethylene glycol
- NH₂-PEG5-NH₂
Fields of Interest: bioconjugation, crosslinking, and materials science
Background & Applications:
Background
3,6,9,12,15-Pentaoxaheptadecan-1,17-diamine (also known as PEG5 diamine, CAS 72236-26-1) is a symmetrical, bifunctional amine-terminated PEG derivative based on a PEG5 backbone, featuring primary amine groups at both termini. The polyethylene glycol spacer provides hydrophilicity, flexibility, and compatibility with aqueous and biological environments while offering extended linker length for increased spatial separation. The terminal primary amines enable efficient coupling to activated carboxylic acids, NHS esters, isocyanates, and related electrophiles. This diamine PEG serves as a versatile building block within a comprehensive portfolio of functionalized PEGs designed for controlled molecular assembly and crosslinking.
Applications
3,6,9,12,15-Pentaoxaheptadecan-1,17-diamine is commonly used in bioconjugation, crosslinking, and materials science applications where dual amine reactivity and extended PEG spacing are required. Typical uses include preparation of PEG-based networks, stepwise construction of multifunctional linkers, PEGylation of polymers and biomolecules, and surface or nanoparticle modification. The presence of two primary amines enables efficient formation of amide or urea linkages, making this compound especially useful in polymer and biomaterials synthesis. As part of a robust functionalized PEG product line, this PEG5 diamine supports modular design strategies in pharmaceutical research, diagnostics, and advanced materials development.
Appearance: Off-white solid
Purity: 98%
Storage: -20 °C
Solubility: Dichloromethane, Chloroform, Methanol
Literature:
- Bioorganic and Medicinal Chemistry, 2009, vol. 17, # 10, p. 3528 – 3535
- Tetrahedron Letters, 1992, vol. 33, # 23, p. 3347 – 3350
- Journal of the American Chemical Society, 2000, vol. 122, # 6, p. 1200 – 1204