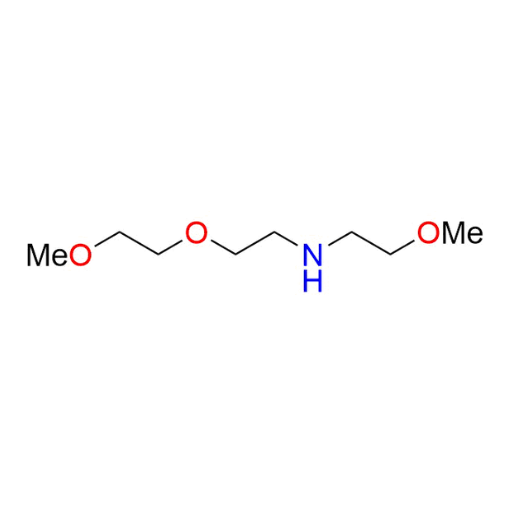
Name: 2-Methoxy-N-(2-(2-methoxyethoxy)ethyl)ethan-1-amine
Molecular Formula: C8H19NO3
CAS#: 128620-95-1
SMILES: COCCOCCNCCOC
MDL#: MFCD16116806
Catalog#: AMTGC527-MA19
Molecular weight: 177.24 g/mol
Other names:
- N-(2-(2-methoxyethoxy)ethyl)-2-methoxyethanamine
- Methoxy-PEG3 secondary amine
- PEG3 dimethoxy amine
Fields of Interest: bioconjugation, materials science, and drug delivery
Background & Applications:
Background
2-Methoxy-N-(2-(2-methoxyethoxy)ethyl)ethan-1-amine is a monofunctional amine-functionalized PEG derivative based on a PEG3 backbone, featuring a terminal secondary amine and methoxy-capped ends. The polyethylene glycol chain provides hydrophilicity, flexibility, and compatibility with aqueous and organic environments, while the methoxy termini remain non-reactive to enable controlled, single-point functionalization. The secondary amine offers tunable reactivity and basicity compared to primary amines, allowing selective transformations and compatibility with a range of electrophiles. This compound serves as a versatile intermediate within a comprehensive portfolio of functionalized PEGs designed for predictable and controlled molecular modification.
Applications
2-Methoxy-N-(2-(2-methoxyethoxy)ethyl)ethan-1-amine is commonly used in bioconjugation, materials science, and drug delivery applications where compact PEG spacing and controlled amine reactivity are required. Typical uses include PEGylation of small molecules and polymers to improve solubility and stability, incorporation into cationic or ionizable systems, and preparation of intermediates for pharmaceutical and biomaterials research. The methoxy-capped PEG chain ensures monofunctional behavior, while the secondary amine allows for selective coupling or further functionalization. As part of a robust functionalized PEG product line, this compound supports modular design strategies in specialty chemical synthesis, diagnostics, and advanced materials development.
Appearance: Colorless liquid
Purity: 98%
Storage: Room Temp
Solubility: Dichloromethane, Chloroform, Methanol
Literature:
- Journal of Medicinal Chemistry, 1991, vol. 34, # 6, p. 1805 – 1818
- Journal of Medicinal Chemistry, 1994, vol. 37, # 2, p. 240 – 247