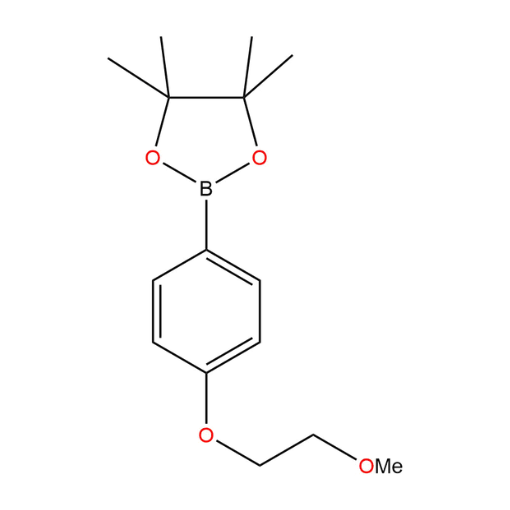
Name: 2-(4-(2-methoxyethoxy)phenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
Molecular Formula: C15H23BO4
CAS#: 959972-40-8
SMILES: CC1(C)OB(OC1(C)C)C2=CC=C(OCCOC)C=C2
MDL#: MFCD23143157
Catalog#: AMTB580-TM12
Molecular weight: 278.15 g/mol
Other names:
- 4-(2-Methoxyethoxy)phenylboronic acid pinacol ester
- Methoxy-PEG2 phenyl boronic ester
- PEG2 aryl boronate linker
Fields of Interest: cross-coupling chemistry, medicinal chemistry, materials science
Background & Applications:
Background
2-(4-(2-methoxyethoxy)phenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (CAS 959972-40-8) is a boronic ester-functionalized aromatic PEG derivative featuring an aryl boronic acid pinacol ester linked to a short methoxy-terminated PEG (PEG2-like) chain. The polyethylene glycol segment provides hydrophilicity, flexibility, and improved solubility in both aqueous and organic systems, while the boronic ester functionality serves as a stable and efficient partner for Suzuki–Miyaura cross-coupling reactions. Compared to boronic acids, pinacol boronate esters offer enhanced stability and ease of handling, making them widely used intermediates in modern synthetic chemistry. This compound represents a versatile building block within a broader portfolio of functionalized PEGs designed for modular assembly and carbon–carbon bond formation.
Applications
This PEGylated aryl boronic ester is commonly used in cross-coupling chemistry, medicinal chemistry, and materials science applications where improved solubility and Suzuki-compatible reactivity are desired. Typical uses include palladium-catalyzed coupling with aryl or vinyl halides to construct substituted aromatic systems, synthesis of PEG-modified pharmaceutical intermediates, and preparation of functional polymers or advanced materials. The presence of a methoxy-capped PEG chain enables controlled, monofunctional incorporation of PEG into target molecules, reducing non-specific interactions and improving physicochemical properties. As part of a comprehensive functionalized PEG product line, this compound supports modular design strategies in drug discovery, diagnostics, and specialty chemical synthesis.
Appearance: Colorless to pale yellow solid
Purity: 98%
Storage: Room Temp
Solubility: Dichloromethane, Chloroform, Acetonitrile
Literature:
- HUTCHISON MEDIPHARMA - WO2012/167423, 2012, A1