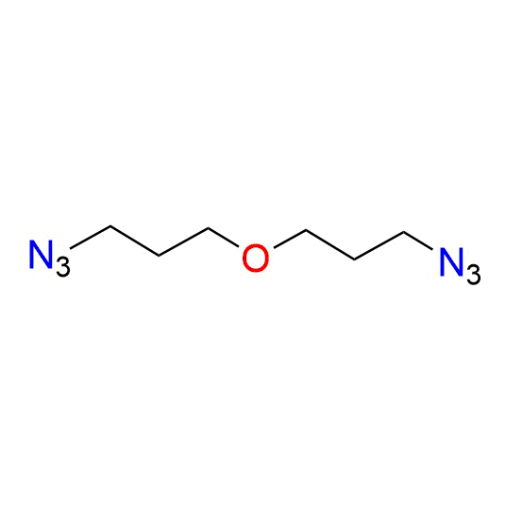
Name: 1-Azido-3-(3-azidopropoxy)propane
Molecular Formula: C6H12N6O
CAS#: 99115-82-9
SMILES: [N-]=[N+]=NCCCOCCCN=[N+]=[N-]
MDL#:
Catalog#: AMTGC907-AA21
Molecular weight: 184.2 g/mol
Other names:
- bis-(3-Azido-propyl)-ether
- 3-(3-Azidopropoxy)propyl azide
- Diazide ether linker
- Bis-azide propyl linker
Fields of Interest: crosslinking, bioconjugation, and materials science
Background & Applications:
Background
1-Azido-3-(3-azidopropoxy)propane (CAS 99115-82-9) is a compact, flexible diazide-functionalized ether linker featuring azide groups at both termini connected through a short propylene glycol-like spacer. While not a traditional PEG chain, the ether linkage provides enhanced flexibility and moderate polarity compared to purely aliphatic linkers. The terminal azide groups enable highly efficient bioorthogonal click chemistry, including copper-catalyzed azide–alkyne cycloaddition (CuAAC) and strain-promoted reactions, allowing simultaneous or stepwise functionalization at both ends. This bifunctional molecule serves as a versatile building block within a broader portfolio of functionalized linkers designed for controlled molecular coupling and crosslinking.
Applications
1-Azido-3-(3-azidopropoxy)propane is commonly used in crosslinking, bioconjugation, and materials science applications where dual azide reactivity and short linker length are desired. Typical uses include construction of polymer networks, preparation of multifunctional intermediates, and surface or nanoparticle modification via click chemistry. Its compact structure allows tight spatial control while maintaining sufficient flexibility for efficient coupling. As part of a comprehensive functionalized linker product line, this diazide linker supports modular design strategies in pharmaceutical research, diagnostics, and advanced materials development.
Appearance: yellow liquid
Purity: 99%
Storage: Room Temp
Solubility: Dichloromethane, Chloroform, Methanol
Literature:
- Bulletin of the Korean Chemical Society, 2024, vol. 45, # 7, p. 639 – 644
- Journal of the American Chemical Society, 1957, vol. 79, p. 3491