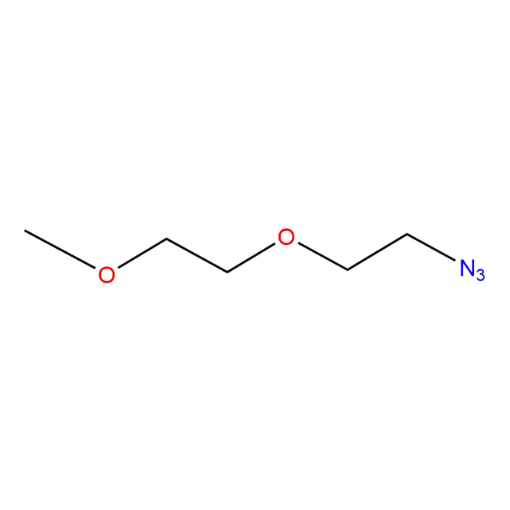
Name: 1-Azido-2-(2-methoxyethoxy)ethane
Molecular Formula: C5H11NO2
CAS#: 215181-61-6
SMILES: COCCOCCN=[N+]=[N-]
MDL#: MFCD19672689
Catalog#: AMTGC225-AZ16
Molecular weight: 145.16 g/mol
Other names:
- 2-(2-Methoxyethoxy)ethyl azide
- Methoxy-PEG2-azide
- PEG2 azide methyl ether
Fields of Interest: bioconjugation, surface modification, and materials science
Background & Applications:
Background
1-Azido-2-(2-methoxyethoxy)ethane is a monofunctional azide-functionalized PEG derivative based on a PEG2 backbone, featuring a terminal azide group and a methoxy-capped end. The polyethylene glycol spacer provides hydrophilicity, flexibility, and compatibility with aqueous and biological environments, while the methoxy terminus remains non-reactive to enable controlled, single-point functionalization. The azide functionality supports efficient bioorthogonal click chemistry, including copper-catalyzed azide–alkyne cycloaddition (CuAAC) and strain-promoted reactions. This compound serves as a versatile intermediate within a comprehensive portfolio of functionalized PEGs designed for predictable and selective molecular modification.
Applications
1-Azido-2-(2-methoxyethoxy)ethane is commonly used in bioconjugation, surface modification, and materials science applications where compact PEG spacing and monofunctional azide reactivity are required. Typical uses include click-based attachment of PEG chains to small molecules, polymers, or surfaces to improve solubility and reduce non-specific interactions, as well as preparation of PEGylated probes and intermediates for diagnostics and imaging. The methoxy-capped PEG chain ensures controlled incorporation of a single reactive site. As part of a robust functionalized PEG product line, this compound supports modular design strategies in pharmaceutical research, biomaterials development, and specialty chemical synthesis.
Appearance: Dark-orange liquid
Purity: 97%
Storage: –20 °C
Solubility: Dichloromethane, Chloroform, Methanol
Literature:
- Macromolecules, 2012, vol. 45, # 12, p. 5151 – 5156
- Journal of the American Chemical Society, 1994, vol. 116, # 12, p. 5057 – 5062