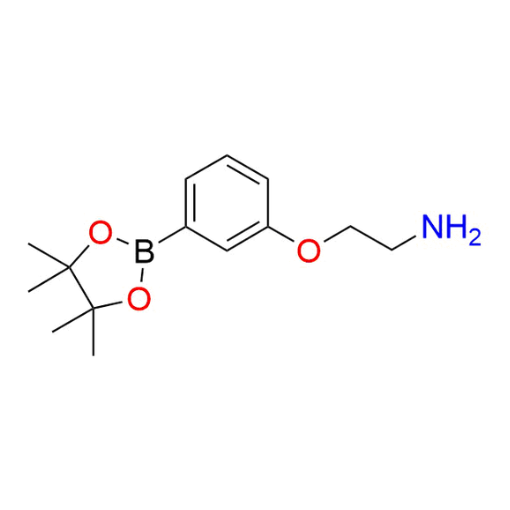
Name: 2-(3-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)ethan-1-amine
Molecular Formula: C14H22BNO3
CAS#: 2684271-71-2
SMILES: CC(C(C)(C)O1)(C)OB1C2=CC=CC(OCCN)=C2
MDL#:
Catalog#: AMTB1432-TD24
Molecular weight: 263.14 g/mol
Other names:
- 2-(3-(Pinacol boronate)phenoxy)ethylamine
- Aryl boronic ester amine linker
Fields of Interest: cross-coupling, medicinal chemistry, materials science, conjugation chemistry
Background & Applications:
Background
2-(3-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)ethan-1-amine (CAS# 2684271-71-2) is a heterobifunctional boronic ester-functionalized linker featuring an aryl boronic acid pinacol ester and a terminal primary amine connected through an ether spacer. The boronic ester moiety provides a stable and reactive handle for Suzuki–Miyaura cross-coupling reactions, while the primary amine enables straightforward coupling to activated carboxylic acids, NHS esters, and isocyanates. The ether linkage contributes flexibility and compatibility with both aqueous and organic systems. This dual-functional architecture makes the compound a versatile intermediate within a broader portfolio of functionalized linkers designed for modular molecular assembly and carbon–carbon bond formation.
Applications
This aryl boronic ester–amine linker is commonly used in cross-coupling chemistry, medicinal chemistry, and materials science applications where orthogonal reactivity is required. It is especially strong as a dual-reactivity building block bridging cross-coupling and conjugation chemistry. Typical uses include palladium-catalyzed coupling with aryl or vinyl halides to construct substituted aromatic systems, followed by amine-based conjugation to peptides, polymers, or small molecules. The combination of Suzuki-compatible boronate chemistry and amine coupling functionality enables stepwise synthesis of multifunctional intermediates, PEGylated analogs, and advanced materials. As part of a comprehensive functionalized linker product line, this compound supports modular design strategies in pharmaceutical research, diagnostics, and specialty chemical synthesis.
Appearance: White Powder
Purity: 98%
Storage: Room Temp
Solubility: Dichloromethane, Chloroform, Acetonitrile
Literature:
- WO2021/155004, 2021, A1Citation 2
![Molecular structure for PEG 4,4,5,5-Tetramethyl-2-[4-(3,6,9,12-tetraoxatridec-1-yloxy)phenyl]-1,3,2-dioxaborolane, also known as Ethoxy-PEG4 phenyl boronic ester (CAS 1181210-11-6), from Frontier Specialty Chemicals.](https://frontierspecialtychemicals.com/wp-content/uploads/2026/03/AMTB1292-TE19-Square-510x510-1.png)