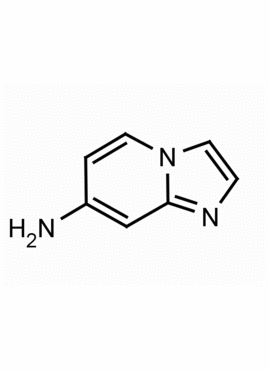
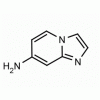
Imidazo[1,2-a]pyridin-7-amine CAS: 421595-81-5 MDL: MFCD11052644
Molecular weight: 133.15 g/mol
Molecular Formula: C7H7N3
CAS Number: 421595-81-5
Storage: Store at 2-8 Co, under dry conditions.
Synonyms: IMIDAZO[1,2-A]PYRIDIN-7-AMINE, 421595-81-5, 7-Aminoimidazo[1,2-a]pyridine, IMIDAZO[1,2-A]PYRIDIN-7-YLAMINE, 7-Amino-imidazo[1,2-a]pyridine, IMIDAZO[1,2-A]PYRIDIN-7-AMINE (9CI), MFCD11052644, SCHEMBL174229
Uses: Nitrogen heterocycle, synthetic building block, reactive amine, synthesis of fine chemicals
Imidazo[1,2-a]pyridin-7-amine, is a synthetic fine chemical useful in the synthesis pf pharmaceuticals, consumer chemicals and photophysical compounds.
Selected References:
Chezal, Jean M., Emmanuel Moreau, Olivier Chavignon, Vincent Gaumet, Jacques Metin, Yves Blache, Anna Diez, Xavier Fradera, Javier Luque, and Jean C. Teulade. Aminoimidazo[1,2-a]Pyridines: Regioselective Synthesis of Substituted Imidazonaphthyridines, Azacarbolines, and Cyclazines. Tetrahedron 58, (2002): 295–307. https://doi.org/10.1016/S0040-4020(01)01141-3.
Fan, Mengyang, Wei Zhou, Yongwen Jiang, and Dawei. Ma. Assembly of Primary (Hetero)Arylamines via CuI/Oxalic Diamide-Catalyzed Coupling of Aryl Chlorides and Ammonia. Organic Letters 17, (2015): 5934–37. https://doi.org/10.1021/acs.orglett.5b03230.
Gobbi, Luca C., Henner Knust, Matthias Korner, Michael Honer, Christian Czech, Sara Belli, Dieter Muri, et al. Identification of Three Novel Radiotracers for Imaging Aggregated Tau in Alzheimer’s Disease with Positron Emission Tomography. Journal of Medicinal Chemistry 60, (2017): 7350–70.
Identification of Three Novel Radiotracers for Imaging Aggregated Tau in Alzheimer’s Disease with Positron Emission Tomography. Journal of Medicinal Chemistry 60, (2017): 7350–70. https://doi.org/10.1021/acs.jmedchem.7b00632.
Walker, Daniel P., F. Christopher Bi, Amit S. Kalgutkar, Jonathan N. Bauman, Sabrina X. Zhao, John R. Soglia, Gary E. Aspnes, et al. Trifluoromethylpyrimidine-Based Inhibitors of Proline-Rich Tyrosine Kinase 2 (PYK2): Structure-Activity Relationships and Strategies for the Elimination of Reactive Metabolite Formation. Bioorganic & Medicinal Chemistry Letters 18, (2008): 6071–77. https://doi.org/10.1016/j.bmcl.2008.10.030.